Note: Descriptions are shown in the official language in which they were submitted.
WO 2022/056573
PCT/AU2021/000024
The title of the invention: INCOMPLETE COMBUSTION AS A MEANS OF
REDUCING CARBON DIOXIDE EMISSIONS
Background of the Invention
[001] Carbon dioxide (CO2) has a significant impact on global warming.
[002] Emissions of Carbon dioxide (C0z) arise from a number of sources, mainly
fossil
fuel combustion to generate energy. Fossil fuel comprises Coal and
hydrocarbons.
[003] Emissions of Carbon dioxide (CO2) arise from complete combustion of
fossil fuel,
according to the following equation:
Fossil Fuel + Oxygen(02) ¨> Carbon dioxide (CO2) + Water (H20) + Flue Gas
Mixture
The source of oxygen for this reaction is the atmospheric air.
[004] Carbon monoxide (CO) is produced instead of Carbon dioxide (CO2) in
incomplete
combustion. Incomplete combustion happens when there is a limited supply of
air,
according to the following equation:
Fossil Fuel + Oxygen(02) ¨> Carbon monoxide (CO) + Water (H20) + Carbon(C)
[005] Water splitting is the chemical reaction in which water is broken down
into oxygen
and hydrogen, according to the following equation:
2 H20 ¨> 2 H2 02
It is, in other words, electrolysis of water or the decomposition of water (1-
120) into
oxygen (02) and hydrogen (H2), due to an electric current being passed through
it.
Production of hydrogen from water is energy intensive. In low-temperature
electrolysis,
the energy consumed is more valuable than the hydrogen produced. Therefore,
this
process is not usually considered commercially viable. I Iowever, with high-
temperature
electrolysis (HTE) of water, more of the initial heat energy is converted into
chemical
energy (hydrogen), thereby improving the commercial viability of this process.
The
commercial viability of this process is further improved by use of renewable
energy
sources to generate electricity for electrolysis.
[006] The use of renewable energy sources in the electrolysis of water
provides means of
exploiting these renewable energy sources. It also provides means of storing
intermittent
renewable power by conversion to chemical energy in hydrogen_
[007] The Fischer-Tropsch is a reaction involving carbon monoxide (CO) and
hydrogen
(H2) to produce clean fuels and other chemical solvents.
1
SUBSTITUTE SHEET (RULE 26) RO/AU
CA 03185797 2023- 1- 11
WO 2022/056573
PCT/AU2021/000024
Detailed Description of the Invention
[001] This invention transforms fuels such as coal and other hydrocarbons from
"dirty" to
:'clean" source of energy. Future power plants may use fossil fuels or the so-
called dirty
fuels to generate Clean energy by using this invention. Existing power plants
that
currently use fossil fuels as feedstock can be amended to commence generation
of Clean
energy by using this invention.
[002] This invention provides the means of harnessing renewable energy
sources. It is
also a viable solution to the problem of intermittent renewable power storage.
[003] This invention provides a "clean" source of cheap commercial scale
synthesis gas,
a fuel gas mixture consisting primarily of carbon monoxide (CO) and hydrogen
(H2).
The fuel gas mixture is used to produce hydrocarbons in a Fischer-Tropsch
process.
[004] These together with additional objects, features and advantages of the
incomplete
combustion as a means of reducing Carbon Dioxide emissions will be readily
apparent to
those of ordinary skill in the art upon reading the following detailed
description of the
presently preferred, but nonetheless illustrative, embodiments when taken in
conjunction
with the accompanying drawings.
[005] In this respect, before explaining the current embodiments of the
incomplete
combustion as a means of reducing Carbon Dioxide emissions in detail, it is to
be
understood that the incomplete combustion as a means of reducing Carbon
Dioxide
emissions is not limited in its applications to the details of design,
construction and
arrangements of the components set forth in the following description. Those
skilled in
the art will appreciate that the concept of this disclosure may be readily
utilized as a basis
for the design of other processes, structures, methods, and systems for
carrying out the
several purposes of the incomplete combustion as a means of reducing Carbon
Dioxide
emissions. It is therefore important that the claims be regarded as including
such
equivalent construction insofar as they do not depart from the spirit and
scope of the
incomplete combustion as a means of reducing Carbon Dioxide emissions. It is
also to be
understood that the phraseology and terminology employed herein are for
purposes of
description and should not be regarded as limiting.
[006] The following detailed description is merely exemplary in nature and is
not
intended to limit the described embodiments of the application and uses of the
described
embodiments_ As used herein, the word "exemplary" or "illustrative" means
"serving as
2
SUBSTITUTE SHEET (RULE 26) RO/AU
CA 03185797 2023- 1- 11
WO 2022/056573
PCT/AU2021/000024
an example, instance, or illustration." Any implementation described herein as
"exemplary" or "illustrative" is not necessarily to be construed as preferred
or
advantageous over other implementations. All of the implementations described
below
arc exemplary implementations provided to enable persons skilled in the art to
practice
the disclosure and are not intended to limit the scope of the appended claims.
Furthermore, there is no intention to be bound by any expressed or implied
theory
presented in the preceding technical field, background or the following
detailed
description.
[007] This invention provides a viable solution to the problem of global
warming as
caused by Carbon dioxide (CO2) emissions arising from fossil fuel combustion
to
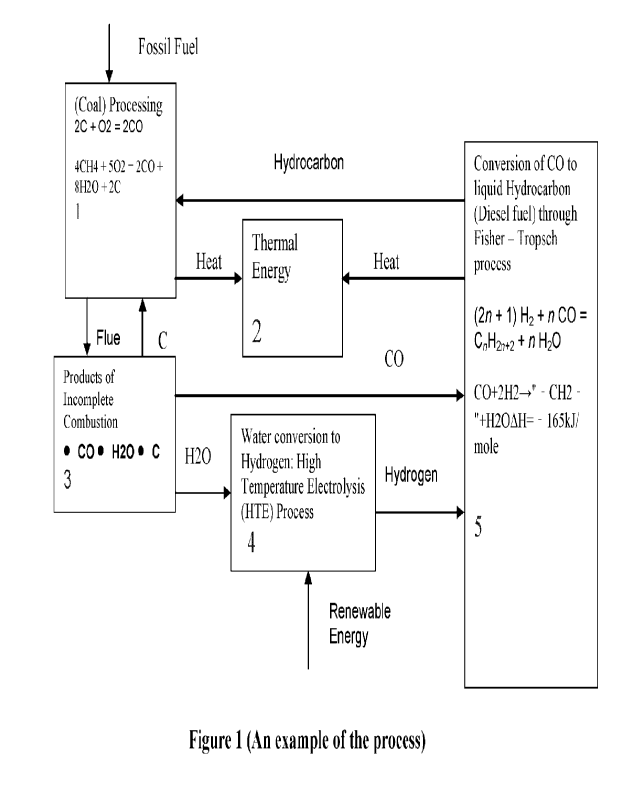
generate energy. In step 1 of the process, this invention uses incomplete
combustion to
ensure an output of carbon monoxide (CO) and water (H20), instead of Carbon
dioxide
(CO2) and Water (F120), as would have been the case if complete combustion
were used.
[008] In step 3 of the process, carbon monoxide (CO), water (H20) and carbon
(C) are
separated. Carbon monoxide (CO), a valuable component of synthesis is sent to
step 5
where it takes part in Fischer-Tropsch reaction, in which fossil fuel in the
form of
hydrocarbon is produced. Carbon (C) is recycled to step 1, while water (H20)
is sent to
step 4, where it is used as feedstock for high-temperature electrolysis (HTE).
[009] In step 4 of the process, water (H20) from step 3 is made to undergo
high-
temperature electrolysis (ME), by using renewable power. This high-temperature
electrolysis (HTE) results in the generation of hydrogen (H2). The use of
renewable
power in this step to generate hydrogen (H2) translates to conversion of these
renewable
energy supply to chemical energy. Hydrogen (H2) is a valuable component of
synthesis
gas and will be used in step 5, along with Carbon monoxide (CO) to create
fossil fuel, in
the form of hydrocarbons. Conversion of renewable energy to chemical energy
provides a
viable solution to the problem of intermittent renewable power storage.
[010] Renewable energy sources include Biofuel, Biomass, Geothermal,
Hydroelectricity,
Solar, Wind, Tidal and Wave. Some of these energy sources are known to produce
intermittent power.
[011] This invention results in the production of synthesis gas, a fuel gas
mixture
consisting primarily of carbon monoxide (CO) and hydrogen (H2), in steps 1 and
4,
respectively. These two gases (carbon monoxide (CO) and hydrogen (H2)) are
made to
3
SUBSTITUTE SHEET (RULE 26) RO/AU
CA 03185797 2023- 1- 11
WO 2022/056573
PCT/AU2021/000024
undergo the Fischer-Tropsch reaction in step 5, to produce clean fuels and
other chemical
solvents. Some of the resulting fuels is recycled to step 1.
[012] According to Vvrikipedia, the Fischer¨Tropsch process is a collection of
chemical
reactions that converts a mixture of carbon monoxide and hydrogen into liquid
hydrocarbons. These reactions occur in the presence of metal catalysts,
typically at
temperatures of 150-300 C (302-572 F) and pressures of one to several tens
of
atmospheres. The process was first developed by Franz Fischer and Hans Tropsch
at the
Kaiser-Wilhelm-Institut fur Kohlenforschung in Germany, in 1925.
[013]With respect to the above description, it is to be realized that the
optimum
dimensional relationship for the various components of the invention described
above
and in following drawing include variations in size, materials, shape, form,
function, and
manner of operation, assembly and use, are deemed readily apparent and obvious
to one
skilled in the art, and all equivalent relationships to those illustrated in
the drawings and
described in the specification are intended to be encompassed by the
invention.
[014] It shall be noted that those skilled in the art will readily recognize
numerous
adaptations and modifications which can be made to the various embodiments of
the
present invention which will result in an improved invention, yet all of which
will fall
within the spirit and scope of the present invention as defined in the
following claims.
Accordingly, the invention is to be limited only by the scope of the following
claims and
their equivalents.
BRIEF DESCRIPTION OF DRAWINGS
The accompanying drawing, which is included to provide a further understanding
of the
invention is incorporated in and constitute a part of this specification,
illustrate an
embodiment of the invention and together with the description serve to explain
the
principles of the invention. They are meant to be exemplary illustrations
provided to
enable persons skilled in the art to practice the disclosure and are not
intended to limit the
scope of the appended claims.
4
SUBSTITUTE SHEET (RULE 26) RO/AU
CA 03185797 2023- 1- 11